The US Food and Drug Administration (FDA) has clarified that the toxic cough syrups linked to the deaths of several children in India were not exported to the United States. The statement comes amid growing international concern over repeated cases of diethylene glycol (DEG) and ethylene glycol (EG) contamination in paediatric medicines produced in India.
🧪 FDA Responds to Global Concern
In an official statement on Friday, the FDA said it was “aware of recent news reports” regarding contamination in children’s cough and cold medicines sold in India.
“The US FDA has confirmed that these specific cough syrups were not shipped to or distributed in the United States,” the agency said.
The regulator added that it continues to work closely with India’s Central Drugs Standard Control Organization (CDSCO) and other global health agencies to ensure contaminated medicines do not enter the US supply chain.
⚠️ WHO Flags Regulatory Gaps
The World Health Organization (WHO) earlier highlighted that India’s drug oversight system has “regulatory gaps” when it comes to testing and screening of locally distributed liquid medicines.
Since 2022, WHO has issued multiple global alerts linking Indian-manufactured cough syrups to child deaths in The Gambia, Uzbekistan, and Cameroon — all of which involved the same toxic compounds, DEG and EG, known to cause kidney failure and severe metabolic acidosis.
🇮🇳 India Bans Toxic Syrups After 17 Child Deaths
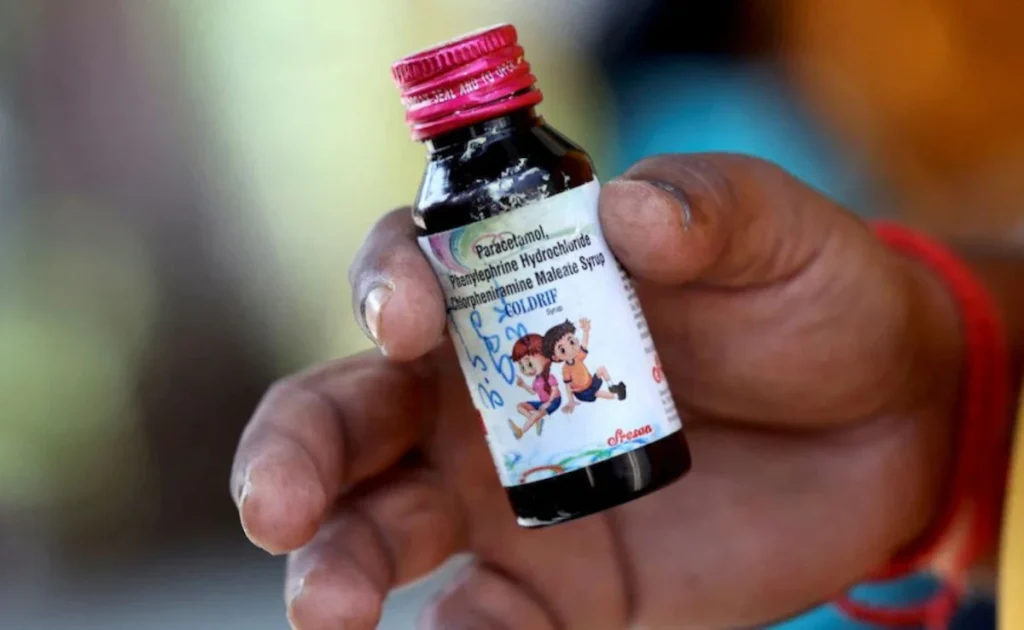
The latest deaths in India — 17 children under the age of five — were reported over the past month and linked to Coldrif, a brand of cough syrup found to contain diethylene glycol at levels nearly 500 times the permissible limit, according to state health authorities.
India’s drug controller ordered an immediate ban on Coldrif after laboratory analysis confirmed contamination on October 2. Two additional cough syrup brands were also flagged for potential toxicity, prompting a nationwide advisory urging parents and pharmacists to avoid untested formulations.
🔍 “Products Not Exported,” Confirms Indian Regulator
The CDSCO informed the US FDA that the toxic syrups in question were “not exported from India to any country.” The confirmation comes as New Delhi faces heightened global scrutiny over pharmaceutical manufacturing standards and export monitoring.
“We remain vigilant and continue to engage with Indian regulators to prevent contaminated products from entering the US market,” the FDA said, urging all drug manufacturers to maintain “the highest safety and quality standards.”
🧫 What Are Diethylene Glycol & Ethylene Glycol?
Both DEG and EG are toxic industrial solvents that can cause acute kidney failure, central nervous system damage, and death if ingested.
These chemicals are sometimes mistakenly used in place of pharmaceutical-grade glycerin — a safe ingredient used to sweeten and stabilize syrups — due to poor quality control and adulteration during manufacturing.
⚙️ Background: India’s Pharma Under Scrutiny
India, one of the world’s largest drug exporters, has faced a series of global alerts since 2022 regarding contaminated cough syrups traced to small and medium-scale manufacturers.
While the government has strengthened rules under the Drugs and Cosmetics Act, experts say more frequent testing, traceability audits, and public transparency are needed to restore global trust in Indian generics.
📊 Summary at a Glance
| Key Point | Details |
|---|---|
| Incident | Child deaths in India linked to toxic cough syrups |
| Chemical Agents | Diethylene glycol (DEG) & Ethylene glycol (EG) |
| Children Dead | 17 (under age 5) |
| Syrup Brand | Coldrif |
| Action Taken | Product banned by Indian authorities |
| US FDA’s Statement | Toxic syrups not shipped or distributed in the US |
| WHO’s Observation | Regulatory gaps in India’s drug testing system |
| Next Steps | Ongoing coordination between FDA, WHO, and CDSCO |









